Tokyo District Court Judgment of October 29, 2025 (Reiwa 7 (Wa) No. 70139)
Case in which the applicability of unfair competition (Article 2, Paragraph 1, Item 21 of the Unfair Competition Prevention Act) was challenged in relation to patent linkage
1. Overview of the incident
The defendant (Bayer Yakuhin) published a notice in an industry newspaper (hereinafter referred to as the “public notice”) and responded to the Ministry of Health, Labour and Welfare (hereinafter referred to as the “MHLW”) (hereinafter referred to as the “Response”), disseminating and announcing that the plaintiff’s (Nihon Generic) manufacturing and sales of generic medical drugs infringed the patent in question (Patent No. 4143297).
In this case, the plaintiff claimed that the defendant’s publication of a public notice in an industry newspaper and its response to the MHLW constituted unfair competition (“disseminating or announcing false facts”) as defined in Article 2, Paragraph 1, Item 21 of the Unfair Competition Prevention Act (hereinafter referred to as the “Unfair Competition Prevention Act”), and sought an injunction under Article 3, Paragraph 1 of the Act and measures to restore credibility under Article 14 of the Act.
The Tokyo District Court dismissed the plaintiff’s claim on the grounds that the public notice and the reply in question could not be said to “harm the business reputation of others” (Article 2, Paragraph 1, Item 21 of the Unfair Competition Prevention Act).
2. Background of the incident
Since April 2012, the defendant has been manufacturing and selling regular rivaroxaban tablets, as well as rivaroxaban OD tablets and granule packets (hereinafter referred to as the “defendant’s products, etc.”).
Under these circumstances, the defendant published a public notice titled “Regarding the Patent Rights Concerning Rivaroxaban” in the industry newspaper Nikkan Yakugyo six times between December 4, 2020, and July 29, 2024.
In February 2023, the plaintiff applied to the Minister of Health, Labour and Welfare for approval to manufacture and sell generic medical drugs (hereinafter referred to as “generic drugs”) such as the defendant’s products as stipulated in Article 14, Paragraph 1 of the Pharmaceuticals and Medical Device Act, and in August 2024, the plaintiff received approval for the manufacture and sale of the above-mentioned approval application, to the extent that the “efficacy or effect” was limited to “inhibiting the onset of ischemic stroke and systemic embolism in patients with non-valvular atrial fibrillation.”
The MHLW and the Pharmaceuticals and Medical Devices Agency (PMDA, collectively referred to as the “MHLW, etc.”) collect information on substance patents or use patents relating to active ingredients of approved prescription drugs (hereinafter referred to as “original drugs”) to ensure a stable supply of generic drugs when conducting approval reviews for generic drugs under the Pharmaceutical and Medical Device Act, and confirm whether or not the generic drugs conflict with the patents of the original drugs. During this process, the MHLW requested explanations from the defendants, who are the patent holders, and received a response (hereinafter referred to as the “Response in this case”).
The plaintiff filed a lawsuit in the Tokyo District Court, claiming that the publication of the public notice in question disseminated “false facts that harm the business reputation of others” (Issue 1) and that the response in question announced, “false facts that harm the business reputation of others” (Issue 2).
3. Relationship with patent linkage
In conducting approval reviews for generic drugs under the Pharmaceutical and Medical Device Act, the MHLW, etc. collect information on substance patents or use patents related to the active ingredients of original drugs and check whether there are any conflicts with the patents of the original drugs in order to ensure a stable supply of generic drugs, based on MHLW notifications (“Regarding the Handling of Pharmaceutical Patent Information Related to Approval Reviews” (Yakushin Notification No. 762, issued by the Director of the Evaluation Division, Pharmaceutical Affairs Bureau, MHLW, October 4, 1994; hereinafter referred to as the “1994 Notification”) and “Regarding the Handling of Pharmaceutical Patents Related to Approval Reviews and Drug Price Listings for Generic Drugs under the Pharmaceutical Affairs Act” (MHLW Notification No. 0605001 and MHLW Notification No. 0605014, issued by the Director of the Economic Affairs Division, Health Policy Bureau, MHLW, and the Director of the Evaluation and Licensing Division, Pharmaceutical and Food Safety Bureau, MHLW, hereinafter referred to as the “2009 Notification”). This practice, which is mandatory for TPP signatory countries, is also known as “patent linkage.” Furthermore, the 1994 and 2009 notifications issued by the MHLW, etc. require patent holders (applicants) of substance patents or use patents (excluding those whose patent term has expired) relating to the active ingredients of original drugs, or persons who have obtained approval for drugs using ingredients covered by such patents as active ingredients (hereinafter referred to as “original drug patent holders, etc.”), to fill out the necessary information on a Drug Patent Information Report Form and submit it to the PMDA. However, submission of the Drug Patent Information Report Form by original drug patent holders, etc. is voluntary, and it will not be made public.
During the approval review process for the plaintiff’s product, the MHLW requested explanations from the defendant, the patent holder of the original drug, based on the above-mentioned notifications. The reply in this case is the defendant’s response to the request.
4. The patent right and its term extension registration
A. The patent right
The patent holder of this patent is a German corporation belonging to the Bayer Group.
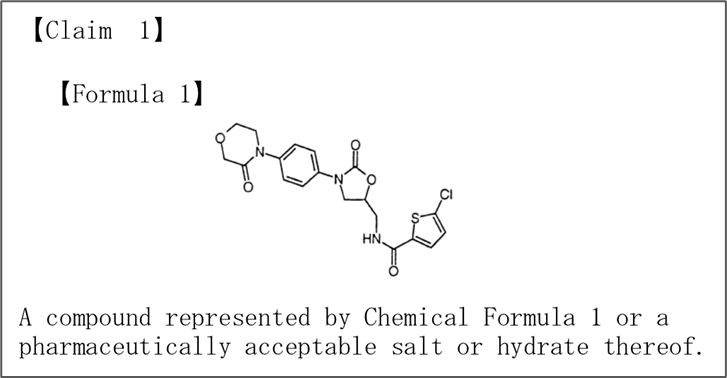
The scope of the claims of this patent is the compound shown in Claim 1 [Chemical Formula 1], sometimes referred to as “rivaroxaban.”
(a) Registration of extension of the term of the patent right
The patent right in question “has had 10 applications for extension of the patent term filed, and those extensions were granted between March 21, 2013 and March 10, 2021 (hereinafter, each extension will be referred to as “Extension Registration 1” etc., corresponding to the number in the attached document, and collectively referred to as “Extension Registrations in Question.” Furthermore, the Act on Ensuring Quality, Efficacy and Safety of Pharmaceuticals, Medical Devices, etc. will be referred to as the “Pharmaceutical and Medical Device Act” throughout, except when cited in relevant laws and regulations.)” and the extension registrations in question “are for pharmaceuticals containing rivaroxaban as an active ingredient (hereinafter, sometimes referred to as “ordinary rivaroxaban tablets” etc., corresponding to the dosage form) that are subject to disposition prescribed by Cabinet Order under Article 67, Paragraph 2 of the Patent Act.”
Of these, the term of the extension registrations (Extension Registrations 3 and 4) for ordinary tablets with the specified use of “treatment and prevention of recurrence of deep vein thrombosis and pulmonary thromboembolism” (hereinafter referred to as “Use 1”) will expire on December 11, 2025.
On the other hand, the court stated that “the terms of the remaining extension registrations, which include Use 1 in the specified uses (i) the extension registration for the fine granule sachets whose specified uses are Use 1 in the specified use (Extension Registrations 7 and 8 in the specified use), and (ii) the extension registration for the OD tablets whose specified uses are Use 1 and “prevention of ischemic stroke and systemic embolism in patients with non-valvular atrial fibrillation” (hereinafter referred to as “Use 2 in the specified use” (Extension Registrations 9 and 10 in the specified use)) expired before February 2023, when the application for manufacturing and selling the plaintiff’s product was filed.”
5. Court Decisions
A. Regarding Issue 1 (whether the publication of the public notice in this case disseminated “false facts that harm the business reputation of others”)
The court stated, “The gist of this public notice is that: 1) the defendant holds five patents, including this patent, that protect products containing rivaroxaban as an active ingredient, and for three of these, including this patent, extensions have been obtained or applications are pending based on the approval of each product, and the defendant intends to take strict and rigorous legal action against any acts that infringe or may infringe these patents (including the parts related to the extension of their duration); and 2) companies that are planning to manufacture, sell, or import products containing rivaroxaban as an active ingredient should take great care to avoid infringing the defendant’s intellectual property rights. The public notice not only does not mention the plaintiff’s products, but also does not mention the dosage form, use, or other components of the products that could be infringing.” It also stated, “The readers of this public notice, which was published in an industry journal, are recognized to be people involved in the pharmaceutical industry, including fellow pharmaceutical companies. Given that the term of the patent right in question (originally set to December 11, 2020) was extended by the extension registration in this case, it is considered that providing information and warnings to fellow pharmaceutical companies by publishing this public notice with the above content in an industry journal was a natural response for an original pharmaceutical manufacturer in order to prevent patent infringement through the manufacture and sale of generic drugs.”
The court then ruled that, “Based on the reader’s ordinary attention and reading, the public notice in this case should be understood as providing information or warning by generally stating that the defendant intends to exercise its rights against any acts that infringe the patent right related to rivaroxaban, based on the obvious premise that the manufacturing and sales of generic drugs containing rivaroxaban as an active ingredient may infringe the patent right of the original drug depending on their composition, including dosage form and use. It is difficult to consider that the public notice in this case should be understood as expressing a view on the scope of the patent right in relation to specific generic drugs, including the plaintiff’s product.” The court ruled that the public notice in this case could not be said to “harm the business reputation of others” (Article 2, Paragraph 1, Item 21 of the Unfair Competition Prevention Act).
B. Regarding Issue 2 (whether the response in question conveys “false facts that harm the business reputation of others”)
The court ruled that “goodwill” in Article 2, Paragraph 1, Item 21 of the Unfair Competition Prevention Act “can be interpreted as a social assessment of the economic value of a business in the trading community, which may influence the decision to do business with that business.” The court also stated that approval for the manufacture and sale of pharmaceuticals is “an administrative disposition made by the Minister of Health, Labour and Welfare based on the authority and responsibility granted by the Pharmaceuticals and Medical Devices Act as a necessary regulation to ensure the quality, efficacy and safety of pharmaceuticals, etc., and to prevent the occurrence and spread of health and hygiene hazards resulting from their use, and is different in nature from transactions in a trading community where free competition takes place.” It also stated that “when reviewing the approval of generic drugs, the MHLW, etc. requests for supplementary explanations from the patent holder of the original drug, etc., as necessary, to confirm whether there is any patent conflict between the original drug and the generic drug, as an act of information gathering prior to an administrative disposition, and in this case, the MHLW, etc. requests for supplementary explanations from the patent holder of the original drug, etc. … as necessary, to confirm whether there is any patent conflict between the original drug and the generic drug.” The court ruled that providing information through this response could not be deemed to “harm the business reputation of others” (Article 2, Paragraph 1, Item 21 of the Unfair Competition Prevention Act) for the following reasons: “It does not appear that the Minister of Health, Labour and Welfare intended to form a public evaluation of the applicant’s business reputation”; and “In the approval review of generic drugs, the Minister of Health, Labour and Welfare makes the decision under his own authority and responsibility, taking into consideration not only the information provided by the patent holder of the original drug but also all other circumstances in a comprehensive manner. Furthermore, since the information provided by the patent holder of the original drug is not to be made public, there is no risk that the information will be leaked to the market and cause a decline in the social evaluation of the applicant’s economic value in the trading community.”
C. Conclusion
The court dismissed the plaintiffs’ claims without ruling on any other issues.
6. Comments
A. Regarding Issue 1 (whether the publication of the public notice in this case disseminated “false facts that harm the business reputation of others”)
In this judgment, the court ruled that the public notice to the industry journal “did not mention the plaintiff’s product” and “did not mention the composition of the product, such as its dosage form or use, which could be considered an infringing product,” and that it “generally stated that the company intends to exercise its rights against acts that infringe patent rights,” based on “the ordinary attention and reading habits of readers,” thus denying that it constituted unfair competition. This judgment, which considers the content of the industry journal and the perspective of its readers in determining whether issuing a warning about the possibility of patent infringement constitutes unfair competition, is useful in practice.
Regarding the illegality of notifying or disseminating information about patent infringement before a court makes a judgment, the Tokyo District Court recently ruled in its judgment on October 9, 2025 (Case No. 70096 (Wa) of 2024 [Main Case] and Case No. 70274 (Wa) of 2024 [Counterclaim]) that, “If the act of notifying or disseminating information that a competitor is infringing intellectual property rights, even though the competitor is not infringing those intellectual property rights, is deemed to have been done as part of the legitimate exercise of intellectual property rights, then, in light of the importance of intellectual property rights, it should be considered to lack illegality.”
B. Regarding Issue 2 (whether the response in question conveys “false facts that harm the business reputation of others”)
In this judgment, the court interpreted “business reputation” as “the social evaluation of a business operator’s economic value in the trading society,” and determined that the administrative disposition by the MHLW “is different in nature from transactions in a trading society where free competition takes place.” The court then ruled that the responses from patent holders of original drugs to the MHLW were “information gathering activities prior to administrative disposition,” and did not “form a social evaluation of the economic value of the applicant for the generic drug,” nor did they “lower the social evaluation regarding economic value” because “the information provided by patent holders of original drugs is not made public.” As a result, the court denied that the responses in this case constituted unfair competition, and this judgment is significant because it shows that appropriate responses to the administration tend not to constitute unfair competition.
The reasoning behind the response in this judgment can be said to be the same as that adopted in the “Eylea case” (Intellectual Property High Court decision of August 13, 2025 (Reiwa 7 (Wa) No. 10003 )). In particular, the idea of placing the provision of information to the government (MHLW, etc.) outside the scope of “defamation” is almost identical. It will be necessary to keep a close eye on future court cases to see whether this kind of thinking will become established.
Furthermore, the patent linkage system is currently being investigated by the MHLW as a research project to explore the possibility of improving its operation and revising the system. It is important to pay attention to future developments regarding revisions to the patent linkage system.